
There may be some overlap between these groups, because there are stronger and weaker π-acceptors, for example, or stronger and weaker σ-donors, but that is the general trend that we would expect. For a given ion, we would expect π-acceptors to give relatively large values of Δ o, whereas σ-donors would give a smaller Δ o and π-donors would give a smaller value still. The configuration of first-row transition metals is also strongly influenced by the ligands in the complex, indicated in Table 3. Note that these examples are compared using complexes with identical ligands. Comparison of Octahedral Field Splitting Between Ions from Different Periods of the Periodic Table 2 ComplexĪlthough the available data is limited here, second-row rhodium has a larger octahedral field splitting than first-row cobalt, and third-row iridium displays a larger field splitting than second-row rhodium. 
First-row transition metal ions, with their smaller Δ o values, are often high spin, but they can also be low spin, and charge is an important factor in determining which case will occur. As a result, transition metal ions from the second and third rows are usually low-spin. The value of Δ o is even larger for third-row transition metals than second-row transition metals. Second, Δ o is much larger for second-row than for first-row metals of the same group, as shown in Table 2. However, comparisons of the field splitting between metals in different columns is complicated, with no simple trend. In each case, the M(III) ion has an octahedral field splitting that is significantly larger than the corresponding value in the M(II) case. Comparison of Octahedral Field Splitting Between M(II) and M(III) Ions 1 Complex These examples are illustrated in Table 1. The higher the charge on an ion, the larger Δ o becomes. First, comparison between Δ o measured for +3 cations and for +2 cations of the first-row transition metals manganese, iron, and cobalt shows that charge exerts a significant influence. It is useful to be aware of some general trends in Δ o. We have already looked at some of the factors that influence the field splitting, so let’s start by looking at that factor. The electron configuration of a d 4 metal ion in an octahedral complex depends broadly on two factors: the difference in energy between the t 2g and e g levels (the octahedral field splitting, Δ o) and the energy associated with pairing two electrons in the same orbital. Having electrons paired in the same orbital leads to a lower spin for the atom. If two spins are pointing in opposite directions, they cancel out, so the overall spin of the atom decreases. If two spins are pointing in the same direction, they add together, so the overall spin of the atom increases. Each electron has a spin of a certain magnitude, but spin is a vector quantity. The terms “high-spin” and “low-spin” really refer to the net spin of the atom. The fourth electron has gone into the higher possible orbital rather than the lower one. It would be at a higher energy level, but it would avoid that repulsive interaction with the other electron in the t 2g orbital. Alternatively, the fourth electron could occupy one of the e g orbitals. The fourth electron has gone into the lower possible orbital rather than the higher possible one. To do so, the fourth electron must be spin-paired with the other occupant of that orbital. The electron may also occupy one of the t 2g orbitals, which lie at lower energy. What about a fourth electron? If the metal ion has a d 4 configuration, we could imagine two situations. 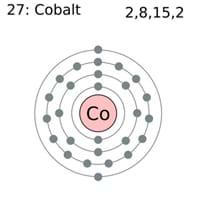
For the d 3 case, one electron will occupy each orbital, with parallel spins. If the complex is octahedral and the metal ion has 1, 2, or 3 d electrons, then the electrons will simply go in the lower level, the t 2g orbitals. Once we have a d orbital splitting diagram for a particular geometry of a complex, we can populate the diagram with the known number of d electrons for a specific metal ion. Exposure to cobalt-60, a powerful gamma ray emitter, may cause cancer.\) Cobalt should be handled with care because of its toxicity and its risk factor in nuclear confrontation. In small amounts, cobalt is an essential element for humans and many other living organisms, and it is also a central component of vitamin B-12 or cobalamin. Brandt between 17 when he was able to show that cobalt colors glass a rich blue. This solid ferromagnetic silver-white element was known in ancient times for its compounds, but its discovery was credited to G. Obtained from: arsenic, oxygen, sulfur, cobatineįrequently, cobalt is associated with nickel because both elements have characteristic ingredients of meteoric iron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
